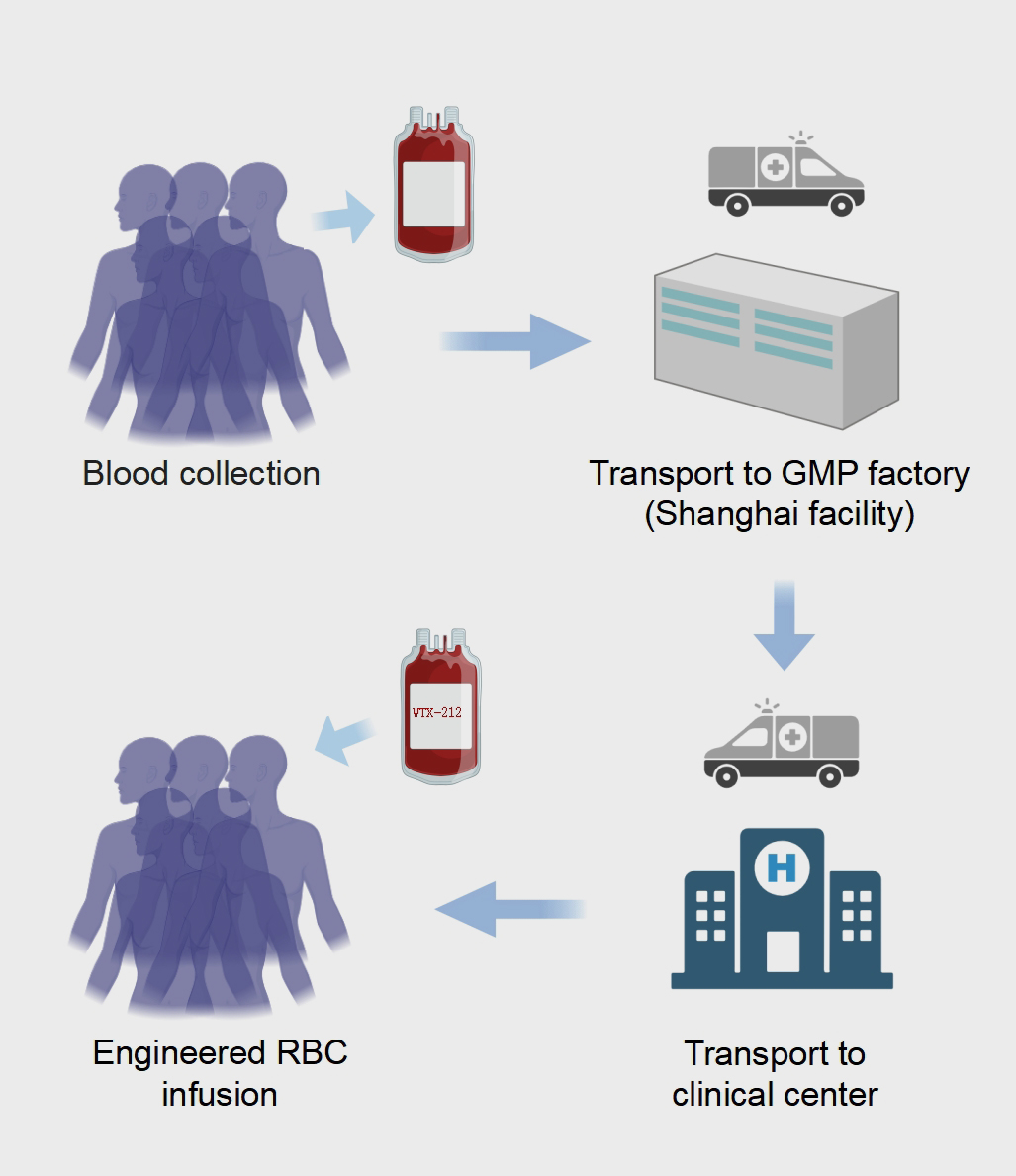
WTX-212: Advanced Solid Tumor Treatment
The study was an investigator-initiated trial designed to evaluate the safety and efficacy of autologous WTX-212 monotherapy in patients with advanced malignancies who had developed resistance to anti-PD-1 therapies. The study was conducted in accordance with all applicable laws, regulations, and guidelines, including the ICH-GCP guidelines and the Declaration of Helsinki. Prior to study enrollment, written informed consent was obtained from all patients. The primary outcome measures included evaluating the safety and tolerability of αPD-1-hEry, measured by the occurrence of DLTs and AEs according to NCI-CTCAE v.5.0. Secondary outcome measures encompassed evaluating the overall Response Rate (ORR), duration of response (DoR), progression-free survival (PFS), and overall survival (OS) according to Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1) criteria, along with assessing the pharmacokinetics of αPD-1-hEry.
CONTACT